|
 |
| jbm > Volume 28(4); 2021 > Article |
|
Abstract
Background
The present study examined the effect of intermittent fasting (IF) on bone mineral content (BMC) and bone mineral density (BMD) and the markers of bone remodeling in a glucocorticoid-induced osteoporosis (GIO) rat model.
Methods
Forty male rats were allocated to 4 groups (N=10 per group): control group of normal rats; control+IF group (normal rats subjected to IF for 16-18 hr daily for 90 days); dexamethasone (DEX) group: (DEX [0.5 mg i.p.] for 90 days); and DEX+IF group (DEX and IF for 90 days). By the end of the experiment, BMD and BMC in the right tibia were measured. Serum levels of the following were measured: glucose; insulin; triglycerides (TGs); total cholesterol; parathyroid hormone (PTH); osteoprotegerin (OPG); receptor activator of nuclear factor-κB (RANK); bone-resorbing cytokines, including bone deoxypyridinoline (DPD), N-terminal telopeptide of collagen type I (NTX-1), and tartrate-resistant acid phosphatase 5b (TRAP-5b); and bone-forming cytokines, including alkaline phosphatase (ALP) and osteocalcin (OC).
Results
DEX administration for 90 days resulted in significantly increased serum levels of glucose, insulin, TGs, cholesterol, PTH, OPG, DPD, NTX-1, and TRAP-5b and significantly decreased BMD, BMC, and serum levels of RANK, OC, and ALP (all P<0.05). IF for 90 days significantly improved all these parameters (all P<0.05).
Glucocorticoids at physiological concentrations have a potentially essential role in the process of bone development and homeostasis.[1] However, administration of glucocorticoids for a long duration results in Cushing disease which has a negative effect on bone health and may be associated with an increased risk of bone fracture.[2] The long-term use of exogenous glucocorticoids as anti-inflammatory or immunosuppressive agents for the management of acute and chronic inflammatory disorders e.g. arthropathies and autoimmune disorders have been increased for last several decades.[3] Glucocorticoid-induced osteoporosis (GIO), a common sequalae for the long-term use of glucocorticoids, is considered the most common secondary cause of osteoporosis.[4] The bone loss caused in GIO is characterized by (1) rapid initial phase during the first year of treatment, which is characterized by loss of bone mineral density (BMD) of around 6% to 12%; and (2) slow long-term phase thereafter, which is characterized by BMD loss of around 3% per year.[5] However, there is a wide heterogeneity in individual susceptibility to the skeletal adverse effects of glucocorticoids.[5]
It has been demonstrated that the process of osteoclastogenesis and bone remodeling involve several signaling pathways such as osteoprotegerin (OPG), receptor activator of nuclear factor-κB (RANK) and RANK ligand (RANKL).[6] Normally, the RANKL stimulates the differentiation and activation of osteoclasts as well as it inhibits the process of apoptosis in these cells, while OPG directly inhibits the binding RANKL with RANK.[6] Also, it has been demonstrated that administration of glucocorticoids significantly increases the expression of RANKL and decreases the expression of OPG in stromal and osteoblastic cells.[7] Moreover, during the process of bone remodeling, several bone-resorbing markers such as N-terminal telopeptide of collagen type I (NTX-1), tartrate-resistant acid phosphatase 5b (TRAP-5b) and deoxypyridinoline (DPD) and bone-forming cytokines such as procollagen type 1 N-terminal propeptide and alkaline phosphatase (ALP), bone specific ALP, or osteocalcin (OC) are released.[8]
Intermittent fasting (IF) is a lifestyle in which you eat for a specific period time, then fast for the rest of the day. Fasting is made possible by the body’s energy storage organs, such as adipose tissue and the liver.[9] IF has been found to lengthen the life, improve energy metabolism, and reduce the chance of acquiring age-related diseases.[10] Furthermore, fasting improves physical and mental performance through enhancing metabolic processes and the nervous system.[11] Experimental evidence demonstrates a preventive role of IF in cognitive decline and many neurodegenerative disorders e.g. Parkinson disease in rodents and human.[12-14] Furthermore, Michalsen and Li [15] found that fasting for 7 to 21 days is efficient for the control of rheumatic diseases, chronic pain syndromes, hypertension, and metabolic syndrome. It has been demonstrated that fasting could improve the bone health by influencing secretion of parathyroid hormone (PTH),[16] which plays a major role in both calcium and phosphate metabolism and the bone remodeling process.[17] Finally, the calorie restriction, a mode of fasting, has been proposed to modulate and potentially slow the progression of age-related diseases such as cardiovascular disease, cancer, obesity, Alzheimer’s disease, and osteoporosis.[18] Based on the results of the previous studies mentioned above, we hypothesized that the IF could improve GIO. Therefore, in the current study, we examined the effect of IF on BMD, bone mineral content (BMC) and serum level of PTH as well as on the bone-resorbing and forming cytokines including OPG, RANK, OC, ALP, NTX-1, TRAP-5b, and DPD which are involved in the process of bone remodeling.
Forty male Sprague-Dawely (SD) rats (12-14 old and 300-350 g) were used in the present study. All rats were housed and kept under controlled conditions (23±1°C; 12 hr light: 12 hr dark cycle) at Nile Center of Experimental Research (NCER), Mansoura, Egypt. All experimental procedures were approved by our local committee of Animal Care (#RP.2018.90). Experiments were performed according to the Laboratory Animals Guide published by the US National Institutes of Health (NIH publication No. 85-23, revised 1996).
The rats were allocated into 4 groups (each 10 rats): (1) control group; included normal male SD rats, fed an ad libitum diet and served as a control group; (2) control+IF group; normal SD rats were fasted daily for 18 hr (14:00-08:00) for 90 days; (3) dexamethasone (DEX) group; SD rats received subcutaneous injections of DEX (0.1 mg/kg/day; Tianjin Pharmaceutical Group Xinzheng Co., Ltd., Zhengzhou, China) for 90 days;[19] and (4) DEX+IF group; SD rats received subcutaneous injections of DEX and exposed to IF as in previous groups.
At the time of sacrifice, blood samples were taken by heart puncture and placed in tubes without anticoagulant for 10 min. To obtain serum, the tubes were centrifuged at 4,000 revolutions per min for 10 min. Until biochemical analysis for the different markers, the serum was kept at −20°C.
All rats’ right hind limbs were dissected from the hip joint after blood sample collection. The BMD and BMC of the right femur and tibia were assessed using a dual energy X-ray absorptiometry scan (LUNAR PIXI #50778; GE Lunar, Madison, WI, USA).[8]
The metaphysis of the left tibia was dissected out, fixed in 10% buffered formalin, and decalcified in ethylenediaminetetraacetic acid solution for 2 weeks. When the bone was decalcified, it was embedded in paraffin. Overall, 5-μm-thick sections were then deparaffinized and stained with hematoxylin and eosin for light microscopic examination.[8]
The concentration of blood glucose was measured by colorimetric method using commercially available kits (Spin React, Girona, Spain) according to manufacturer instructions. Also, serum TGs and total cholesterol were measured by commercially available kits as directed by the manufacturer (Human Diagnostics, Wiesbaden, Germany). In addition, serum insulin was measured by enzyme-linked immunoassay (ELISA) insulin kits for rats (Sun-Red biology and technology, Shanghai #cat no 201-11-0708) as directed by the manufacturer.
ELISA kits were used for measurement of the serum level of ALP (Beyotime, P0321, Shanghai, China), PTH (MBS2700368), OPG (MBS27101236), TRAP-5b (MBS2702692), cross-linked NTX-1 (MBS2700254), DPD (MBS2506789), OC (MBS2701838), and RANK (MBS2704130).[8]
The serum levels of glucose and insulin showed a significant increase in DEX group compared to control and control+IF group (P<0.001). On the other hand, IF+DEX group showed a significant reduction in serum glucose and insulin compared to DEX group (P<0.001) (Fig. 1A, B).
Figure 2 shows that DEX administration for 90 days caused a significant rise in the serum level of cholesterol not the serum TGs compared to control and control+IF group (P< 0.001). To the contrary, the use of IF with DEX caused a significant reduction in serum levels of TGs and total cholesterol compared to DEX group (P<0.001) (Fig. 2A, B).
DEX group had considerably lower BMD and BMC than control and control+IF groups (P<0.0001), whereas IF+DEX group had significantly higher BMD and BMC than DEX group (P<0.05) (Fig. 3A, B).
Histological changes were examined by hematoxylin and eosin staining. The tibiae from the control and control +IF groups demonstrated normal structure and arrangement of the trabeculae (Fig. 4A, B respectively). In the DEX group, the trabeculae become thin and reduced with small numbers of empty bone lacunae (Fig. 4C). These changes were revised in DEX+IF group (Fig. 4D).
The level of PTH was significantly higher in DEX group compared to control and control+IF group (P<0.0001), while its level in IF+DEX group was significantly lower compared to DEX group (P<0.05) (Fig. 5).
When compared to the control and control+IF groups, the serum level of OPG, a RANK inhibitor, was considerably lower in the DEX group (P<0.0001). In comparison to the DEX group, the IF+DEX group demonstrated a significant rise in OPG (P<0.0001) (Fig. 6A). However, when comparing the DEX group to the control and control+IF groups, the serum level of the osteoclastogenic factor; RANK was considerably greater in the DEX group (P<0.0001). In comparison to the DEX group, the IF+DEX group demonstrated a significant rise in RANK (P<0.0001) (Fig. 6B).
DPD, TRAP-5b, and NTX-1 serum levels were considerably elevated in the DEX group compared to the control and control+IF groups (P<0.0001). In comparison to the DEX group, the IF+DEX group demonstrated a substantial drop in their levels (P<0.05) (Fig. 7A-C).
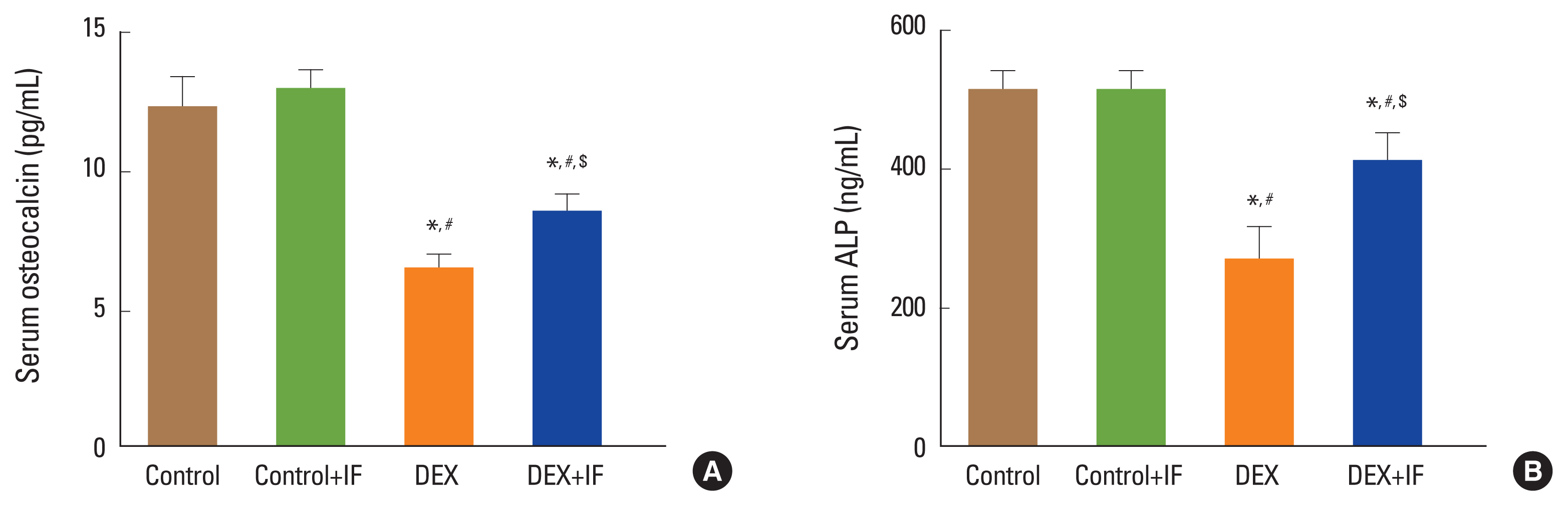
The DEX group had considerably lower serum levels of OC and ALP than the control and control+IF groups (P< 0.0001). In comparison to the DEX group, the IF+DEX group demonstrated a substantial rise in their levels (P<0.05) (Fig. 8A, B).
The main findings of the current work included (1) administration of DEX for 90 days caused significant deteriorations in glucose homeostasis, BMC and BMD, which was associated with significant increase in serum PTH, RANK and bone-resorbing markers (DPD, TRAP-5b, and NTX-1) and a significant reduction in serum level of OPG and bone-forming markers (OC and ALP); and (2) IF for 18 hr/day caused significant improvement in glucose homeostasis, BMD, BMC, and markers of bone turnover.
The first objective of the current study was to examine the effect of IF on the impaired glucose tolerance caused by long-term use of glucocorticoids. The present study demonstrated a significant increase in fasting blood glucose, insulin, TGs and cholesterol, findings confirming previous studies and suggesting impairment of glucose tolerance with the long-term use of glucorticortoids. Moreover, IF caused significant improvement in glucose tolerance. These findings are in agreement with that reported by previous studies. Gotthardt et al. [20] reported that IF increases insulin sensitivity and improves glucose tolerance. In a rodent model of hyperphagic and obese mice due to deficiency of brain-derived neurotrophic factor, alternate day fasting (ADF) reverses insulin resistance, and decreases the circulating levels of insulin.[21] Also, Arum et al. [22] reported that IF significantly improves the glucose regulation/insulin sensitivity in long-lived Ames dwarf mice and growth hormone receptor mutant mice.
The long-term use of glucocorticoids is characterized by impairment in bone formation as evidenced by a reduction in BMC and BMD.[23] In consistence with the previous studies, the current study relieved that the long-term use of DEX for 90 days causes significant reduction in the BMC and BMD with loss of trabeculae suggesting development of osteoporosis. Moreover, IF for 16 to 18 hr/day for 90 days caused a significant improvement in bone mass (BMC and BMD) and morphology suggesting anti-osteoporotic effect for IF in GIO. In agreement with these findings, a human study by Kormi et al. [24] demonstrated that ramadan fasting reduced the development of osteoporosis and Xu et al. [25], in a rodent study, demonstrated that a combination of IF with ketogenic diet prevented the reduction in BMD and bone resorption induced by ketogenic diet. On the other hand, Barnosky et al. [26] demonstrated that an ADF in human showed no significant changes in BMC and BMD and Hisatomi and Kugino [27] demonstrated that single fasting for 96 hr in male Wistar rats caused marked reduction in bone density without any major change in the macroscopic morphology of bone and BMC is restored within 4 days after resumption of feeding.
It has been shown that glucocorticoids can alter PTH secretion directly, upregulate PTH receptors in skeletal muscles, and increase their affinity for PTH, which could improve PTH sensitivity.[5] In line with earlier research, the current study found a significant increase in PTH levels after 90 days of long-term glucocorticoid administration, demonstrating that glucocorticoids may alter PTH secretion. Furthermore, we discovered that IF dramatically reduced high PTH levels, implying that inhibition of PTH secretion may be one of the mechanisms behind IF’s anti-osteoporotic activity in GIO.
The bone is a dynamic structure that continuously undergoes a process of formation and breakdown called bone remodeling. This process of bone remodeling is regulated by the activity of osteoclasts and osteoblasts cells. One of the main mechanisms involved in the process of osteoclastogenesis is the RANK/RANL/OPG pathway. Glucocorticoids alter the RANKL-OPG signaling pathway. With long-term glucocorticoid treatment, the synthesis of RANKL in osteoblasts and osteocytes increases,[7,28,29] but the expression of OPG at the mRNA level decreases.[28] Furthermore, the increase in the ratio of RANKL to OPG caused by glucocorticoids is related to the promotion of osteoclast development and maturation, which leads to increased bone resorption.[5] In line with these findings, the current study found that administering DEX for 90 days caused a significant increase in bone resorption biomarkers and cytokines such as RANK, TRAP-5b, NTX-1, and DPD suggesting that long-term glucocorticoid administration increases osteoclast activity and the development of osteoporosis. Glucocorticoids, on the other hand, generate considerable reductions in OPG (RANK inhibitor), OC, and ALP (markers of osteoblast activity), suggesting that osteoblast activity is suppressed and bone production is reduced.
In addition, the current study found that IF for 16 to 18 hr in rats resulted in a significant increase in serum levels of OPG and bone-forming biomarkers such as OC and ALP, as well as a significant decrease in serum levels of RANK and bone-resorbing biomarkers such as TRAP-5b, NTX-1, and DPD, suggesting that IF can inhibit GIO by inhibiting osteoclast activity and stimulating osteoblast activity. In line with these findings, some controlled clinical trials of caloric restriction found declines in bone formation biomarkers, such as OC,[30] ALP,[31] and increases in bone resorption indicators, such as C-terminal telopeptide of type I collagen.[26,31] On the other hand, Kormi et al. [24] found that fasting during Ramadan prevented the development of osteoporosis by lowering the amount of dipeptidyl peptidase-4.
Limitations of the current study include that we did not measure the serum level of RANKL (we measured RANK which is a reasonable index for the process of osteoclastogenesis) and the number of osteoclasts and osteoblast by histomorphometry study. These points will be considered in future study.
We concluded that long-term use of glucocorticoids in rats caused reduction in BMC, BMD which was associated with a rise in the serum levels of PTH, RANK, DPD, and NTX-1 and reduction in the serum levels of OPG, OC, and ALP. On the other hand, IF for 90 days corrected GIO probably via inhibiting the osteoclastogenesis, stimulating the activity of osteoblasts and inhibiting the secretion of PTH.
Acknowledgments
All authors contributed to all points of the research and nobody can be acknowledged.
DECLARATIONS
Fig. 1
Serum levels of fasting glucose (mg/dL) (A) and fasting insulin (μU/mL) (B) in different groups. Data were expressed as mean±standard deviation. *Significant vs. control group. #Significant vs. control+intermittent fasting (IF) group. $Significant vs. dexamethasone (DEX) group. P ≤ 0.05 is considered significant.
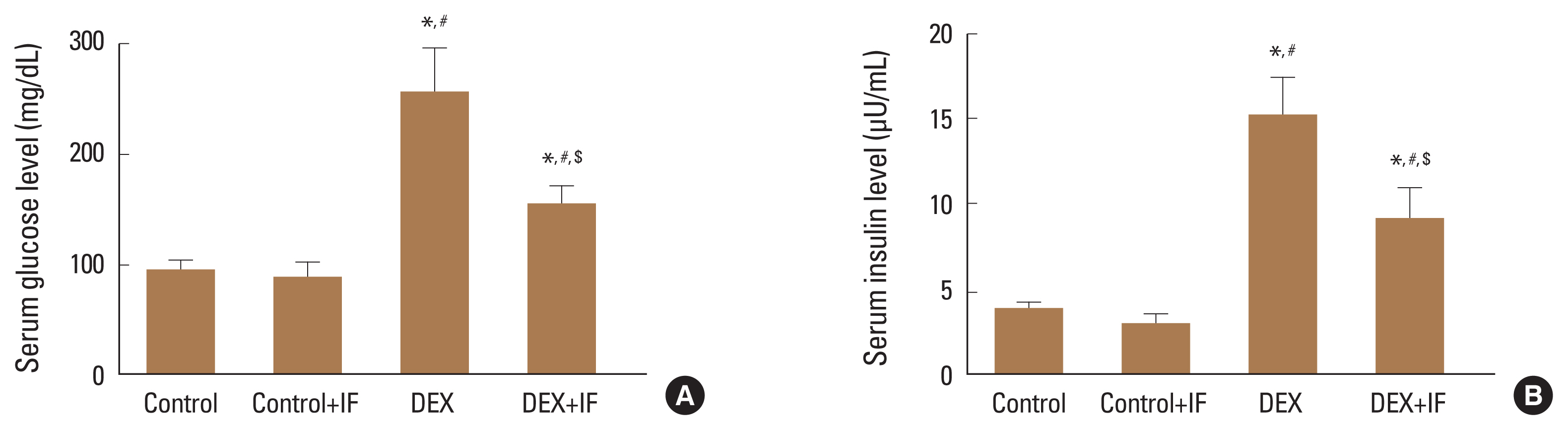
Fig. 2
Serum levels of triglycerides (TGs) (mg/dL) (A) and total cholesterol (mg/dL) (B). Data were expressed as mean±standard deviation. *Significant vs. control group. #Significant vs. control+intermittent fasting (IF) group. $Significant vs. dexamethasone (DEX) group. P≤0.05 is considered significant.
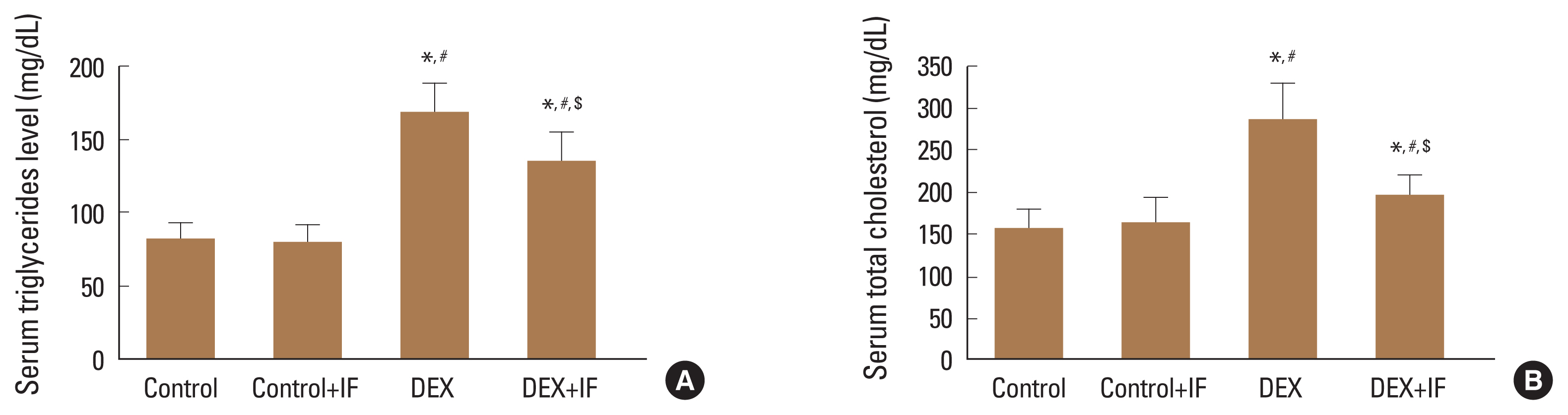
Fig. 3
Bone mineral density (BMD) (A), bone mineral content (BMC) (B) in different groups. Graphs of X-rays pictures from (C) control group, (D) control+intermittent fasting (IF) group, (E) dexamethasone (DEX) group and (F) IF+DEX group. Data were expressed as mean±standard deviation. *Significant vs. control group. #Significant vs. control+IF group. $Significant vs. DEX group. P≤0.05 is considered significant. ROI, region of interest.

Fig. 5
Serum level of parathyroid hormone (PTH) in different studied groups. Data were expressed as mean±standard deviation. *Significant vs. control group. #Significant vs. control+intermittent fasting (IF) group. $Significant vs. dexamethasone (DEX) group. P≤0.05 is considered significant.
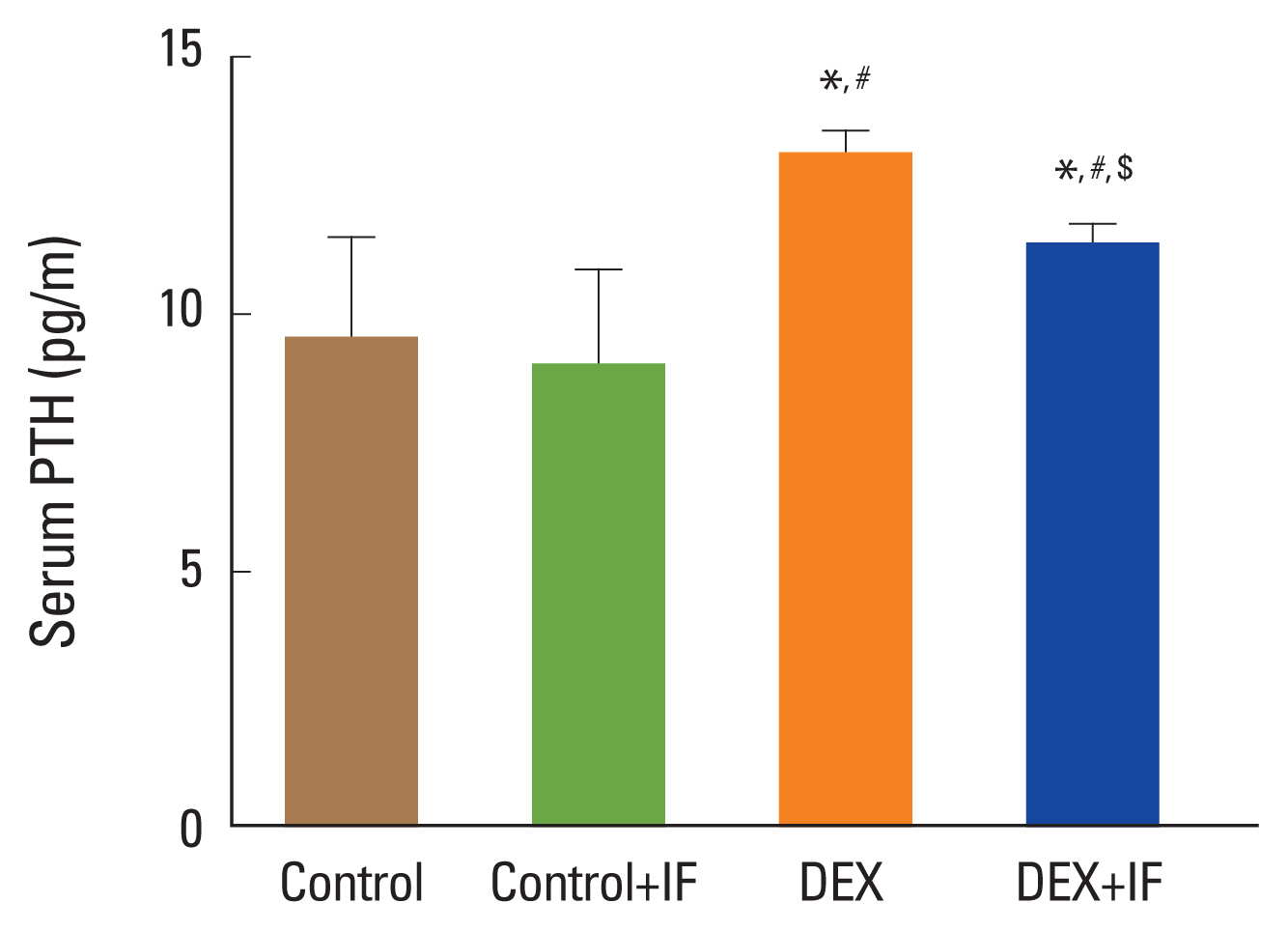
Fig. 4
Histopathological examination of the proximal metaphysis of the tibia using hematoxylin and eosin (H&E) in different groups. The specimens from the control and intermittent fasting (IF)+control groups show a network of cancellous bone trabeculae with osteocytes within their lacunae within bone trabeculae (black arrows) (×200 magnification) (A, B), from the dexamethasone (DEX) group show loss of trabecular bone with bone marrow (BM) spaces and empty lacunae (blue arrows) (×200 magnification) (C), and from the IF+DEX group shows n normal trabecular bone architecture with osteoblasts at endosteal surface (black arrows) (×200 magnification) (D).
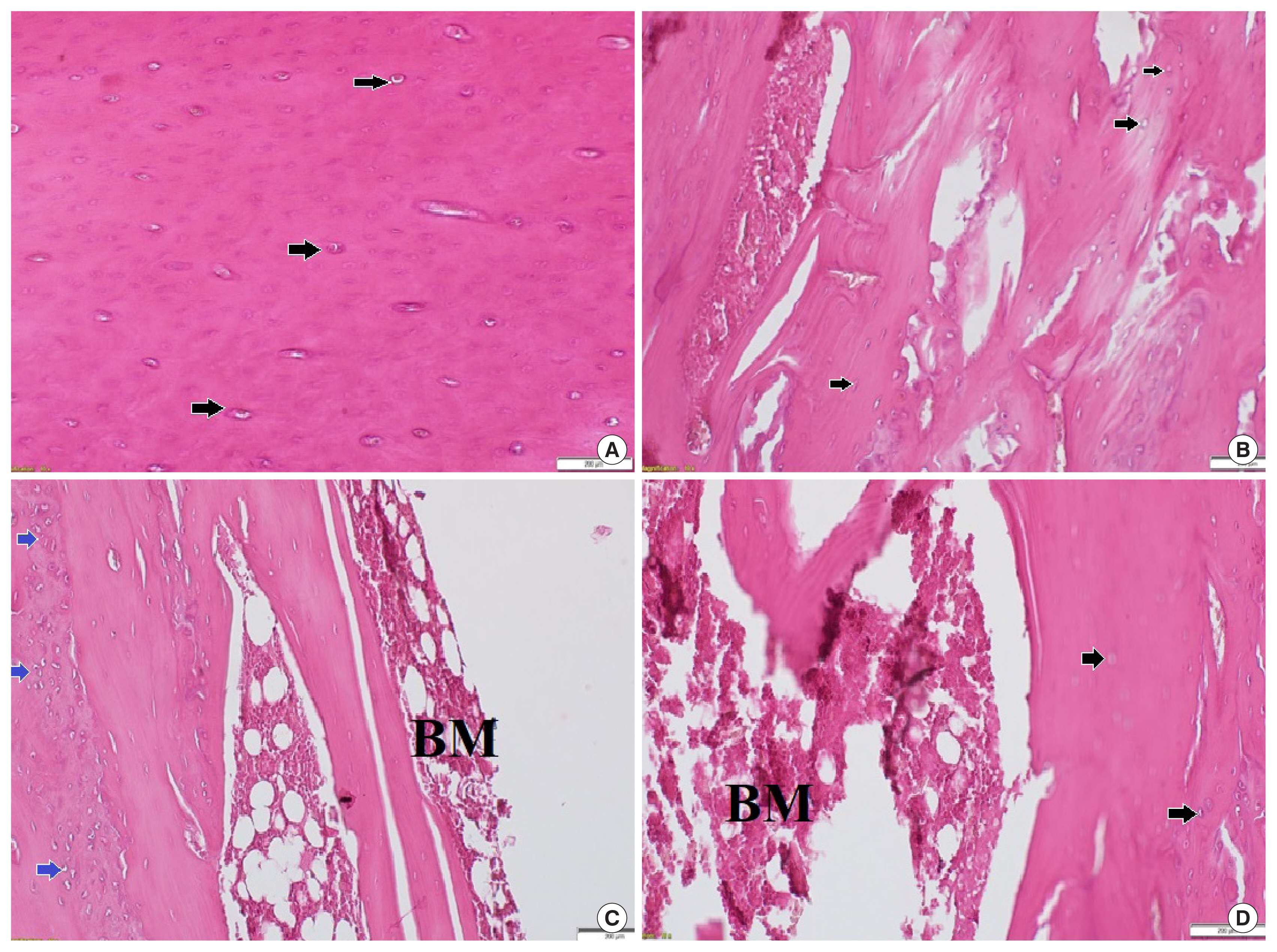
Fig. 6
Serum levels of osteoprotegerin (pg/mL) (A) and receptor activator of nuclear factor-κB (RANK) (ng/dL) (B) in different groups. Data were expressed as mean±standard deviation.*Significant vs. control group. #Significant vs. control+intermittent fasting (IF) group. $Significant vs. dexamethasone (DEX) group. P ≤0.05 is considered significant.
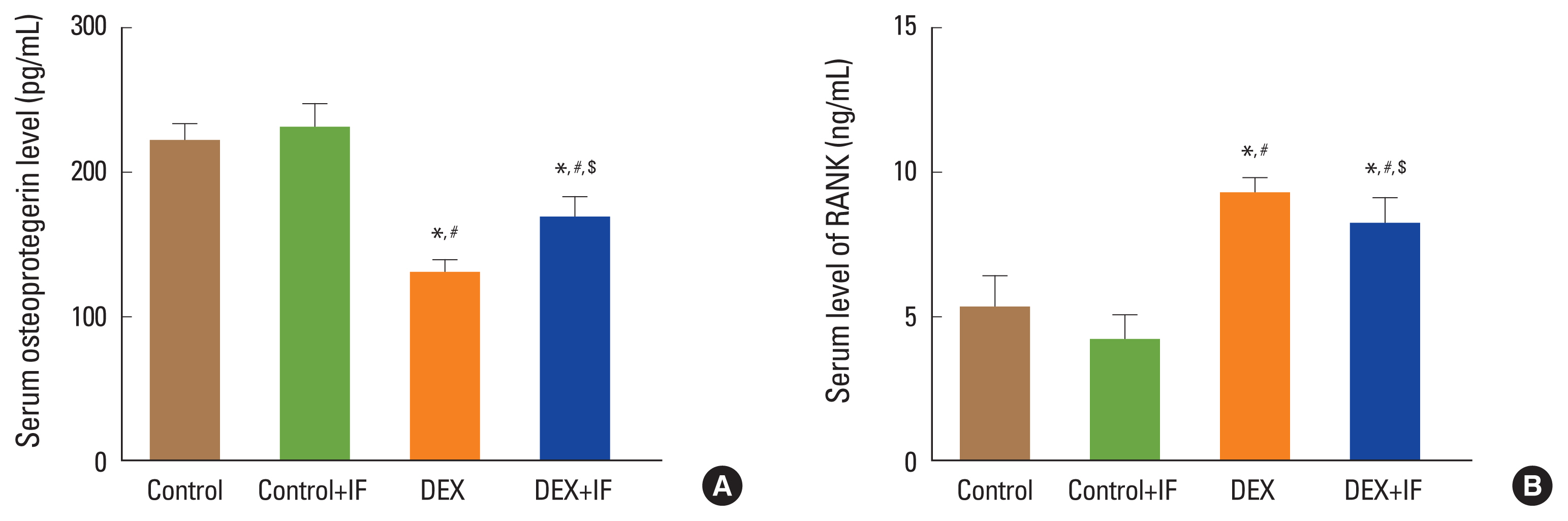
Fig. 7
Serum levels of deoxypyridinoline (DPD) (ng/mL) (A), tartrate-resistant acid phosphatase (TRAP)-5b (ng/mL) (B) and N-terminal telopeptide of collagen type I (NTX-1) (ng/mL) (C) as bone resorbing biomarkers in different groups. Data were expressed as mean±standard deviation. *Significant vs. control group. #Significant vs. control+intermittent fasting (IF) group. $Significant vs. dexamethasone (DEX) group. P ≤0.05 is considered significant.

Fig. 8
Serum levels of osteocalcin (pg/mL) (A) and alkaline phosphatase (ng/mL) (B) in different groups. Data were expressed as mean±standard deviation. *Significant vs. control group. #Significant vs. control+intermittent fasting (IF) group. $Significant vs. dexamethasone (DEX) group. P ≤ 0.05 is considered significant.
REFERENCES
1. Caplan A, Fett N, Rosenbach M, et al. Prevention and management of glucocorticoid-induced side effects: A comprehensive review: Gastrointestinal and endocrinologic side effects. J Am Acad Dermatol 2017;76:11-6. https://doi.org/10.1016/j.jaad.2016.02.1239.


2. Kaltsas G, Makras P. Skeletal diseases in Cushing’s syndrome: osteoporosis versus arthropathy. Neuroendocrinology 2010;92:Suppl 1. 60-4. https://doi.org/10.1159/000314298.


3. Vandewalle J, Luypaert A, De Bosscher K, et al. Therapeutic mechanisms of glucocorticoids. Trends Endocrinol Metab 2018;29:42-54. https://doi.org/10.1016/j.tem.2017.10.010.


4. Compston J. Glucocorticoid-induced osteoporosis: an update. Endocrine 2018;61:7-16. https://doi.org/10.1007/s12020-018-1588-2.



5. Chotiyarnwong P, McCloskey EV. Pathogenesis of glucocorticoid-induced osteoporosis and options for treatment. Nat Rev Endocrinol 2020;16:437-47. https://doi.org/10.1038/s41574-020-0341-0.


6. Kwan Tat S, Padrines M, Théoleyre S, et al. IL-6, RANKL, TNF-alpha/IL-1: interrelations in bone resorption pathophysiology. Cytokine Growth Factor Rev 2004;15:49-60. https://doi.org/10.1016/j.cytogfr.2003.10.005.


7. Swanson C, Lorentzon M, Conaway HH, et al. Glucocorticoid regulation of osteoclast differentiation and expression of receptor activator of nuclear factor-kappaB (NF-kappaB) ligand, osteoprotegerin, and receptor activator of NF-kappaB in mouse calvarial bones. Endocrinology 2006;147:3613-22. https://doi.org/10.1210/en.2005-0717.


8. Sakr HF, Hussein AM, Eid EA, et al. Impact of dehydroepiandrosterone (DHEA) on bone mineral density and bone mineral content in a rat model of male hypogonadism. Vet Sci 2020;7:185.https://doi.org/10.3390/vetsci7040185.


9. Mattson MP, Longo VD, Harvie M. Impact of intermittent fasting on health and disease processes. Ageing Res Rev 2017;39:46-58. https://doi.org/10.1016/j.arr.2016.10.005.


10. Redman LM, Ravussin E. Caloric restriction in humans: impact on physiological, psychological, and behavioral outcomes. Antioxid Redox Signal 2011;14:275-87. https://doi.org/10.1089/ars.2010.3253.



11. Longo VD, Mattson MP. Fasting: molecular mechanisms and clinical applications. Cell Metab 2014;19:181-92. https://doi.org/10.1016/j.cmet.2013.12.008.



12. Hu Y, Yang Y, Zhang M, et al. Intermittent fasting pretreatment prevents cognitive impairment in a rat model of chronic cerebral hypoperfusion. J Nutr 2017;147:1437-45. https://doi.org/10.3945/jn.116.245613.


13. Hu Y, Zhang M, Chen Y, et al. Postoperative intermittent fasting prevents hippocampal oxidative stress and memory deficits in a rat model of chronic cerebral hypoperfusion. Eur J Nutr 2019;58:423-32. https://doi.org/10.1007/s00394-018-1606-4.


14. Zhang J, Zhan Z, Li X, et al. Intermittent fasting protects against Alzheimer’s disease possible through restoring aquaporin-4 polarity. Front Mol Neurosci 2017;10:395.https://doi.org/10.3389/fnmol.2017.00395.



15. Michalsen A, Li C. Fasting therapy for treating and preventing disease - current state of evidence. Forsch Komplementmed 2013;20:444-53. https://doi.org/10.1159/000357765.


16. Bahijri SM, Ajabnoor GM, Borai A, et al. Effect of Ramadan fasting in Saudi Arabia on serum bone profile and immunoglobulins. Ther Adv Endocrinol Metab 2015;6:223-32. https://doi.org/10.1177/2042018815594527.



17. Fraser WD, Logue FC, Christie JP, et al. Alteration of the circadian rhythm of intact parathyroid hormone and serum phosphate in women with established postmenopausal osteoporosis. Osteoporos Int 1998;8:121-6. https://doi.org/10.1007/bf02672507.


18. Martin SL, Hardy TM, Tollefsbol TO. Medicinal chemistry of the epigenetic diet and caloric restriction. Curr Med Chem 2013;20:4050-9. https://doi.org/10.2174/09298673113209990189.



19. Chen Z, Xue J, Shen T, et al. Curcumin alleviates glucocorticoid-induced osteoporosis through the regulation of the Wnt signaling pathway. Int J Mol Med 2016;37:329-38. https://doi.org/10.3892/ijmm.2015.2432.


20. Gotthardt JD, Verpeut JL, Yeomans BL, et al. Intermittent fasting promotes fat loss with lean mass retention, increased hypothalamic norepinephrine content, and increased neuropeptide Y gene expression in diet-induced obese male mice. Endocrinology 2016;157:679-91. https://doi.org/10.1210/en.2015-1622.


21. Duan W, Guo Z, Jiang H, et al. Reversal of behavioral and metabolic abnormalities, and insulin resistance syndrome, by dietary restriction in mice deficient in brain-derived neurotrophic factor. Endocrinology 2003;144:2446-53. https://doi.org/10.1210/en.2002-0113.


22. Arum O, Saleh JK, Boparai RK, et al. Preservation of blood glucose homeostasis in slow-senescing somatotrophism-deficient mice subjected to intermittent fasting begun at middle or old age. Age (Dordr) 2014;36:9651.https://doi.org/10.1007/s11357-014-9651-2.



23. Canalis E, Mazziotti G, Giustina A, et al. Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int 2007;18:1319-28. https://doi.org/10.1007/s00198-007-0394-0.


24. Kormi SMA, Ardehkhani S, Kerachian MA. The effect of islamic fasting in ramadan on osteoporosis. J Nutr Fasting Health 2017;5:74-7. https://doi.org/10.22038/JFH.2017.22955.1086.

25. Xu X, Ding J, Wu X, et al. Bone microstructure and metabolism changes under the combined intervention of ketogenic diet with intermittent fasting: an in vivo study of rats. Exp Anim 2019;68:371-80. https://doi.org/10.1538/expanim.18-0084.



26. Barnosky A, Kroeger CM, Trepanowski JF, et al. Effect of alternate day fasting on markers of bone metabolism: An exploratory analysis of a 6-month randomized controlled trial. Nutr Healthy Aging 2017;4:255-63. https://doi.org/10.3233/nha-170031.



27. Hisatomi Y, Kugino K. Changes in bone density and bone quality caused by single fasting for 96 hours in rats. PeerJ 2019;6:e6161.https://doi.org/10.7717/peerj.6161.



28. Hofbauer LC, Gori F, Riggs BL, et al. Stimulation of osteoprotegerin ligand and inhibition of osteoprotegerin production by glucocorticoids in human osteoblastic lineage cells: potential paracrine mechanisms of glucocorticoid-induced osteoporosis. Endocrinology 1999;140:4382-9. https://doi.org/10.1210/endo.140.10.7034.


29. Piemontese M, Xiong J, Fujiwara Y, et al. Cortical bone loss caused by glucocorticoid excess requires RANKL production by osteocytes and is associated with reduced OPG expression in mice. Am J Physiol Endocrinol Metab 2016;311:E587-93. https://doi.org/10.1152/ajpendo.00219.2016.



30. Villareal DT, Shah K, Banks MR, et al. Effect of weight loss and exercise therapy on bone metabolism and mass in obese older adults: a one-year randomized controlled trial. J Clin Endocrinol Metab 2008;93:2181-7. https://doi.org/10.1210/jc.2007-1473.



31. Villareal DT, Fontana L, Das SK, et al. Effect of two-year caloric restriction on bone metabolism and bone mineral density in non-obese younger adults: A randomized clinical trial. J Bone Miner Res 2016;31:40-51. https://doi.org/10.1002/jbmr.2701.


- TOOLS
-
METRICS

- ORCID iDs
-
Majed G. Alrowaili

https://orcid.org/0000-0002-1869-5205Abdelaziz M. Hussein

https://orcid.org/0000-0002-0239-175XElsayed A. Eid

https://orcid.org/0000-0002-2311-2238Mohamed S. Serria

https://orcid.org/0000-0001-8113-4542Hussein Abdellatif

https://orcid.org/0000-0001-5590-5112Hussein F Sakr

https://orcid.org/0000-0003-3918-3046 - Related articles
-
Prevention and Treatment of Glucocorticoid-induced Osteoporosis1994 April;1(1)




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print