Effect of Cross-Sex Hormones on Body Composition, Bone Mineral Density, and Muscle Strength in Trans Women
Article information
Abstract
Background
Cross-sex hormone therapy (CHT) changes the physical characteristics of transgender women to match their gender identity and expression. This study aimed to determine the effects of feminizing cross-sex hormones on body composition, bone mineral density (BMD) and muscle strength in transgender women.
Methods
A prospective observational study assessed 11 participants who underwent feminizing CHT. Dual energy X-ray absorptiometry (DXA), and handgrip strength were measured before CHT and after 6-months of CHT. Fat mass, lean body mass (LBM), and BMD were measured by DXA and handgrip strength was measured by hand-dynamometer.
Results
Regional body fat in the trunk, legs, and gynoid region increased by 18%, 27.4%, and 27.2%, respectively after 6 months of CHT. Total body fat increased by 16.2%, while the fat mass ratio decreased by 7.2%. Although body fat increased, the android/gynoid fat ratio decreased; BMD in the lumbar spine significantly increased by 3.9% (P=0.0051), but changes in the femoral neck (P=0.1969) and total femur (P=0.4769) were not significant. Changes in LBM ranged from −3% (trunk) to −8% (arm region). Right-hand grip strength also significantly decreased by 7.7% (P=0.0467).
Conclusions
After 6 months of CHT, transgender women showed a general increase in fat mass and a decreased in overall LBM and handgrip strength. Increase in fat mass percentage were more remarkable in gynoid region, leading to a more “female” body fat distribution.
INTRODUCTION
Gender is defined as the attitudes, feelings, and behaviors linked to one’s biological sex’s experience and expression. Gender identity refers to a person’s own experience of who they are and is not the same as sexual orientation, which is related to attraction.[1] In addition, the terms “transgender,” “transsexual,” “trans,” “gender nonbinary,” “gender incongruent,” and “genderqueer” are adjectives for persons with gender identities that are not aligned with the sex recorded at birth, while “cisgender” is the term for a person whose sex recorded at birth aligns with their gender identity.[2]
Transgender people may seek any gender-affirming interventions, including hormone therapy, surgery, facial hair removal, interventions for speech and communication modifications, and behavioral adaptations. These procedures have been defined as medically necessary by the World Professional Association for Transgender Health (WPATH).[3] Social, hormonal, and surgical transitions are associated with improved emotional health and well-being and are now widely viewed as effective treatments for gender dysphoria.
The 2 significant hormone therapy goals aim to reduce endogenous sex hormone levels and secondary sex characteristics of the individual’s designated gender and replace endogenous sex hormone levels consistent with the individual’s gender identity by using principal hormone replacement treatment for hypogonadal patients.[4]
The general approach of feminizing therapy combines estrogen to develop female sex characteristics and androgen blockers to suppress or minimize secondary male characteristics. In comparison, masculinizing hormone therapy involves using several forms of parenteral testosterone to develop male secondary characteristics. This cross-sex hormone therapy (CHT) changes their physical appearance to match their gender identity and expression better.
Sex steroids, particularly estrogen, play a crucial role in attaining and maintaining peak bone density in people. Transgender women, compared to trans men, have been frequently observed to have low bone density before undergoing gender-affirming hormone therapy, and many studies show improved bone density in trans women. Using pharmacologic testosterone, bone density in trans men remains mostly unchanged, although androgens have indirect effects on bone health via fat and lean mass changes.[5]
Sex steroids might have direct and indirect effects on body fat and lean body mass (LBM). Direct effects include estrogen binding to estrogen receptors, which stimulates pre-adipocyte proliferation and lipoprotein lipase activity and promoting body fat storage. In contrast, the binding of testosterone to androgen receptors inhibits lipopolysaccharide activity and increases adipose tissue lipolysis.[6]
After considering these aspects, this study aimed to determine the effects of feminizing cross-sex hormones on bone mineral density (BMD), body fat, and LBM in different body regions of trans women. In addition, the effects on the change of muscle power were evaluated.
METHODS
1. Study population and study design
A prospective observational study was conducted in a gender clinic of a university hospital, and all individuals 19 years old and older diagnosed with gender dysphoria from each psychologist and started feminizing cross-sex hormone between January and December 2019 were eligible for participation in the study. However, those who previously underwent cross-sex hormone use, gonadectomy, or had psychological vulnerability were not eligible. All eligible individuals then visited the outpatient gender clinic every 3 months for hormone therapy, laboratory tests, and general checkups (Fig. 1).
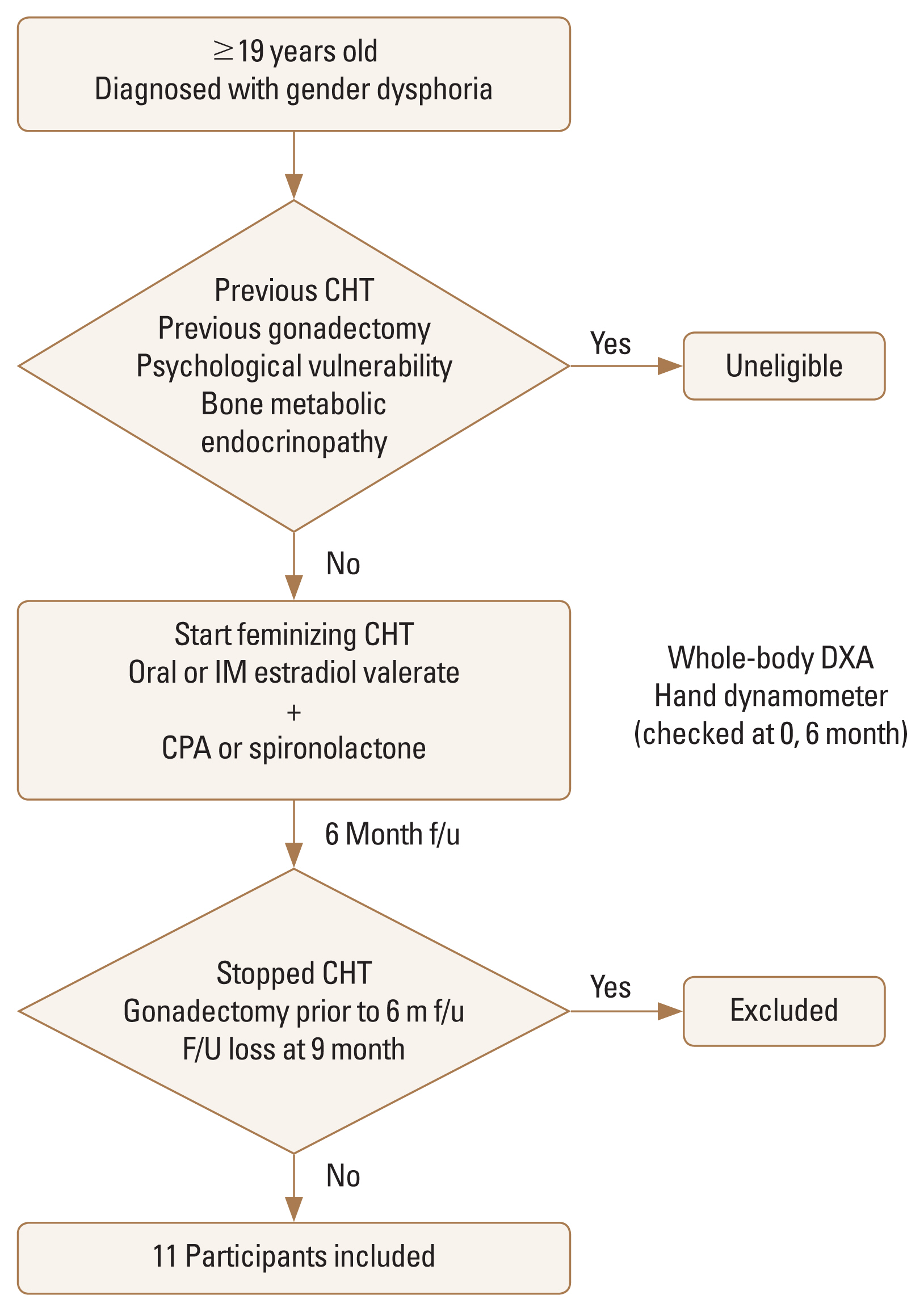
Flow chart of study selection based on the inclusion and exclusion criteria. CHT, cross-sex hormone therapy; CPA, cyproterone acetate; F/U, follow-up; DXA, dual energy X-ray absorptiometry.
Individuals were included for the present analysis if they had 6-month feminizing CHT, underwent whole-body dual energy X-ray absorptiometry (DXA), and a grip strength test with hand dynamometer before undergoing and after 6 months of feminizing CHT. Potential participants were excluded if they stopped hormone therapy or underwent gonadectomy before their 6-month hormone therapy. In addition, persons were excluded if the follow-up DXA or hand dynamometer was not obtained 9 months after the start of the CHT. These inclusions and exclusions resulted in 11 participants for the present analyses.
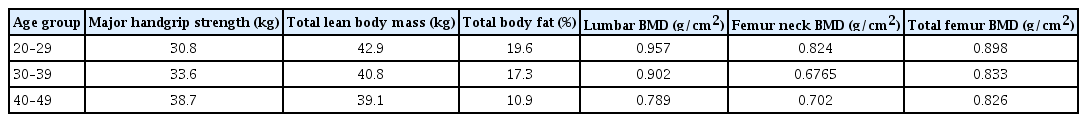
Of the 11 participants, 8 were between ages 20 to 29 years old, 2 were between ages 30 to 39 years old, and one was between ages 40 to 49. The average values of the variables affected by age are shown in Table 1. The computed BMD among participants was compared to the average BMD of women and average BMD men from the same age group in South Korea (Table 2).
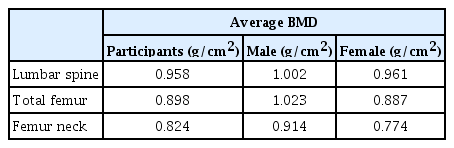
Comparison of BMD between the Korean general population and participants who are in the 20 to 29 age group
This study had Institutional Review Board approval, and all patients provided written informed consent based on the institution’s guidelines.
2. Treatment protocol
Feminizing hormone treatment regimens started after the initial visit. As patients preferred intramuscular (IM) estradiol injection and oral estradiol administration, 11 trans women were treated with oral 2 to 4 mg estradiol valerate (EV) per day or 5 to 10 mg of IM EV every 2 weeks. Both were combined with androgen blocker, using either 25 to 50 mg cyproterone acetate per day or 100 mg spironolactone per day (Table 3). For spironolactone and cyproterone acetate, their price difference provided options for the patients. In addition, if the estradiol levels did not show sufficient elevation in the 6th-month follow-up after the first treatment, the dose was increased.
3. Measurement of bone density and body composition by DXA
Anthropometric data, including weight, height, and body mass index, were assessed during the first visit. All participants underwent DXA (Discovery-W fan-beam densitometer; Hologic Inc., Waltham, MA, USA) to assess BMD and body composition without specific preparation. Their BMD was measured at the lumbar spine (L1–L4), the femoral neck, and the total femur, and the resulting value was expressed in g/cm2. The least significant change (LSC) was also computed to determine if BMD changes were significant (95% confidence interval). The LSC of the total femur and the lumbar spine are 0.027 g/cm2 and 0.022 g/cm2, respectively.
Body fat, LBM, total body mass, and specific region mass, such as the arm, leg, trunk, head, android region, and gynoid region, were measured using DXA. The Hologic Apex software (13.6.05) used in the scan analysis defined the android and gynoid regions. The android region is found around the waist, located between the lumbar spine’s midpoint and the pelvis’s top. Meanwhile, the gynoid region is found between the femur head and the middle of the thigh.[7] Appendicular skeletal muscle mass was calculated as the sum of muscle mass in the arms and legs, assuming that all non-fat and non-bone tissues are skeletal muscle. Skeletal muscle mass index (SMI) was calculated by dividing the limb skeletal muscle mass (kg) by the square of the height (m2).
4. Measurement of grip strength
A Takei T.K.K. 5401 GRIP-D handgrip dynamometer (Takei Scientific Instruments, Niigata, Japan) was used for data collection. Although American Society Hand Therapist (ASHT) recommends using hydraulic type dynamometer (Jamar and its variants), this mechanical type dynamometer (Smedley type) is commonly used in Asia, and its test-retest reliability is known to be excellent.[8] Measurements were performed with the participants standing up, arm at their side with their elbows fully extended. The participants were asked to squeeze the dynamometer thrice with both hands with a 1 min resting period between each set to overcome fatigue. The mean value of three squeezes was computed.
A Wilcoxon signed-rank test was used to compare variables between baseline and 6 months after CHT. Rex (version 4.0.0; RexSoft Inc., Seoul, Korea) was used for analysis, and a P-value less than 0.05 was considered to be statistically significant.
RESULTS
Eight trans women and 3 non-binary individuals with female spectrum gender identity participated in the study. A total of 11 participants had an average age of 29 years ranging from 20 to 47 years of age (Table 3).
Ten of the 11 participants were right-handed, and 1 participant was left-handed. Participants did not lose or gain body weight but lost 4% of their LBM and gained 16.2% body fat (Table 4).
1. Changes in regional body fat
Regional body fat increased in all body parts except the head region (−2.6%), with notable increases in the trunk (17.7%), android region (23.0%), gynoid region (27.2%), and leg (27.4%). Both android and gynoid regions have increased body fat. However, the android/gynoid fat ratio decreased by 5% in 6 months. This increase in regional body fat leads to a significant increase in total body fat by 16.2% and a 7.2% decrease in the fat mass ratio. Despite the increase in estimated visceral fat mass (13.6%), it was not considered significant (P=0.4769) (Table 4).
2. Changes in LBM
Regional changes in LBM ranged from −2.6% (trunk region) to −8% (arm region). The LBM in the trunk and head did not change significantly, but the arm’s and leg’s LBMs decreased significantly, leading to decreased total body LBM by 4.3%. The SMI also decreased significantly by 8.1% (P=0.0076) (Table 4).
3. BMD and handgrip strength
Except for those in their 30s and 40s, who have a small sample size, the initial total femur and femur neck BMDs of the participants in their 20s were lower than males but higher than females of the general population of South Korea (Table 2).[9]
The BMD of the lumbar spine significantly increased by 3.9%, while the BMD changes in the femur neck (+1.2%, P=0.1969) and total femur (+0.7%, P=0.4769) were not significant (Table 4). The right hand’s grip strength significantly decreased by 7.7% (P=0.0467), but the decrease on the left hand’s grip strength is not significant (6.2%, P=0.1688) (Table 4).
DISCUSSION
The changes in body composition after estrogen treatment in those born with a male phenotype have been well documented. Thus, trans women, after long-term CHT, show a general increase in fat and a decrease in overall LBM. To further evaluate the effects of feminizing cross-sex hormones in different body regions of trans women, objective DXA measures were used. Compared to anthropometry used in previous studies to estimate body composition change,[7,8] DXA is a more accurate method. In a previous study that examined gender differences in regional body compositions, comparing regional fat percentages using DXA demonstrated a substantially increased fat mass in women’s limbs but a much smaller increase in the trunk. Thus, the study adjusted the sex differences for body weight and reported the persistence of increased leg fat percentages in women, but a lack of significant differences for the arm and the trunk. Hence, a change of regional body composition is a more appropriate value when adjusting their gender identity.[10] Lapauw et al., [11] also using DXA, found that the total lean mass of trans women was 20% lower, and total fat mass was 30% higher than the male control group. Other studies showed similar results.[12,13] In this study, LBM in the trunk and head did not change significantly, but the decreased LBM in the arm and leg led to a significant decrease in total body LBM. Accordingly, the SMI also decreased significantly by 8.1%. Regional body fat increased in all body parts except the head region, which led to a significant increase in total body fat and a decrease in fat mass ratio. Body fat increased, and the android/gynoid fat ratio decreased for 6 months. Although the estimated visceral fat mass increased, it was not significant. The results showed that in trans women, the increase in body fat percentage is higher in the gynoid region than in the android region, consistent with feminine body fat distribution and similar during puberty. Because of increasing estrogen levels, females develop total fat and store them mainly in the gluteal and femoral regions, leading to a gynoid body shape.[14] These changes mimic natal puberty in that pubertal development was complete in 18 to 24 months after initiating hormone therapy in trans women, which helps them obtain the physical characteristics of the desired sex.
Sex steroids are influential determinants of bone homeostasis in adulthood, and in this study, the BMD was used to examine the effects of CHT on the participants’ bone density. The initial total femur and femur neck BMDs of participants in their twenties were lower than the BMD of the males but higher than the BMD of females in the general population (Table 2).[9] This result is similar to the results of previous studies.[15] The exact reason is still unclear, but some authors had speculated that the initial lower muscle strength of transgender women before the treatment resulted in the lower BMD.[16] In trans women, estrogen therapy seems to maintain bone mineralization homeostasis despite testosterone withdrawal.[17–19] Based on the results, BMD in the lumbar spine increased significantly, while femur neck and total femur changes were not significant. Similarly, some previous studies reported increased bone mass with estrogen therapy performed in less than 2 years.[20,21] Furthermore, systemic reviews and meta-analyses on a total of 392 trans women revealed an increase in BMD at the lumbar spine, but not at the femoral neck, after 12 and 24 months of estrogen therapy.[22] Taken together, data from these studies and the present work suggest that estrogen therapy can maintain bone mass.
The expected anabolic effect of testosterone withdrawal on muscle mass after CHT is reflected in the data. Grip strength significantly decreased in the right hand by 7.7% (P=0.467) but not in the left hand (−6%, P=0.1688). Scharff et al. [23] found that after a year of CHT, grip strength decreased in trans women, which resulted from the lack of testosterone. Testosterone plays an essential role in muscle strength because it affects myoblast proliferation and myoblast differentiation and increases the number of satellite cells, promoting muscle mass protein synthesis.[24] Knowing the effects of CHT on grip strength can help medical providers and trans women with CHT’s physical expectations.
Although this study was limited because its sample size was not large enough to obtain a robust P-value, its contribution to describing CHT’s short-term effect on transgender people is valuable because studies on this topic are scarce in Korea.[25] This study is a preliminary step in producing a study with more participants and long-term follow-up to confirm CHT’s long-term effects on patients and examine whether trans women ultimately obtain their desired sex’s body composition. This study can also help prepare a basis for treatment selection by confirming differences in patients’ reactions and the degree of side effects depending on the type of therapeutic hormone agent. In addition, further long-term studies are expected to determine the relationship of bone mass with body fat and muscle mass.
In recruiting participants for this study, individuals receiving medical treatment for a condition that could affect musculoskeletal health, such as thyroid disease, were excluded. However, most transgender participants were taking antidepressants before CHT. It is currently unclear whether the change of body composition is due to CHT alone or combined with antidepressants. In a further study, it would be required to consider the psychiatric comorbidities or concomitant drugs as baseline characteristics to gauge the pure effect of the CHT.
In conclusion, trans women show a general increase in fat and decreased overall LBM and grip strength after short term CHT. Differences in fat are more remarkable in gynoid fat, resulting in a more feminine body fat distribution.
Notes
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Ministry of Science and ICT (No. 20181031).
Ethics approval and consent to participate
The study protocol conformed to the ethical guidelines of the World Medical Association Declaration of Helsinki and was approved by the Institutional Review Board. Written informed consents were obtained from all participants.
Conflict of interest
No potential conflict of interest relevant to this article was reported.